Cementite is in fact of much wider interest than in metallurgy alone, within subjects spanning from astrophysics, planetary science, Lunar processes, and biomedicine to name but a few. The impact of carbon-containing meteorites with the moon, is speculated to lead to a reduction of the iron-containing minerals on its surface the resulting reaction with the carbonaceous gases generated by the impact to produce cementite (Jull:1975). In mineralogy, the carbide is known as cohenite (Fe,Ni,Co) 3C, after the German mineralogist Emil Cohen, who was investigating material of meteoric origin. The carbide was therefore envisioned to cement the iron. The name has its origins in the theory of Osmond and Werth, in which the structure of solidified steel consists of a kind of cellular tissue, the iron constituting the nucleus and the carbide the envelope of the cells. Structure factor calculations for cementite.Electron diffraction patterns from cementite.Data corresponding to X-ray diffraction pattern of cementite.Its metastability mostly does not matter over the time scale and conditions of normal life. This is because it is hard at ambient temperature, as we shall see, due to its crystal structure that has a much lower symmetry than all the forms in which the iron occurs. Some 50 million tonnes of cementite is produced annually within about 1.6 billion tonnes of steel, adding enormously to the quality of life. All equilibria in this sense are metastable even the constituents of atoms will all decay eventually if the Universe keeps on expanding. Instead of considering just two phases together, if we now put iron, graphite and cementite in mutual contact at ambient pressure then the cementite eventually must give way to the more stable equilibrium between graphite and iron. However, the term is a tenuous concept, because there might be something else also consisting of Fe and C, which may be more stable. Such diagrams identify domains, for example in temperature and composition space, where either a single phase or a combination of phases is stable. It is possible, therefore, to find equilibria between iron and graphite, iron and diamond and iron and cementite, represented conventionally by the respective binary, two-phase diagrams. In its crystalline, liquid and glassy states, iron has an affinity for carbon, whether to form a solution over a wide range of compositions, as graphite or diamond, or in the form of compounds with narrowly defined compositions, such as cementite. Solution to the Bagaryatskii and Isaichev ferrite-cementite orientation relationship problem.Comprehensive review on cementite, containing detailed references to the information presented below.All rights reserved.The structure of cementite Cementite H. 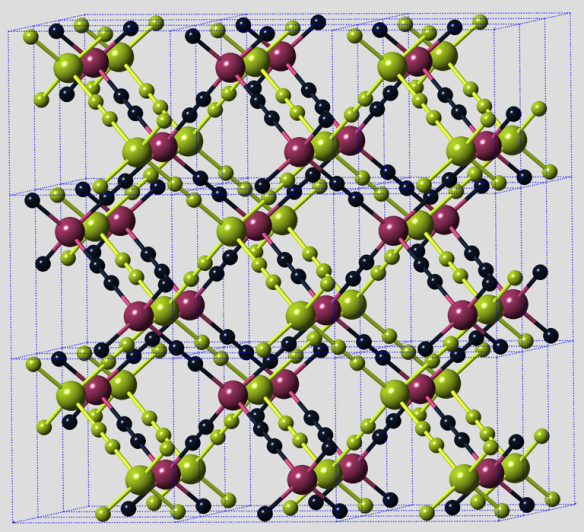
IMPORTANT: You must include your IP address, otherwise we won't know which address to unblock! You can check your actual IP address (as seen by the outside world) using a free service such as Ĭopyright © 2022 CrystalMaker Software Ltd. Please contact us to request that we unblock your access. Have we made a mistake? If you believe we have mistakenly blocked your access, we apologise. You, or another user with the same IP address, has been detected posting spam, attempting to hack this site, or making a denial-of-service attempt. So please purchase the appropriate licence(s) from us, and delete any stolen software from your computer. Without this income there would be no software. We rely on software sales to fund our research and development. You, or another user with the same IP address, has been detected using an illegal, stolen ("hacked") copy of our software.ĭo not use stolen or "hacked"/"cracked" software: it's illegal - and it's seriously uncool: nobody likes a cheat. Access Denied You are forbidden to access this website because: Either:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
